Introduction
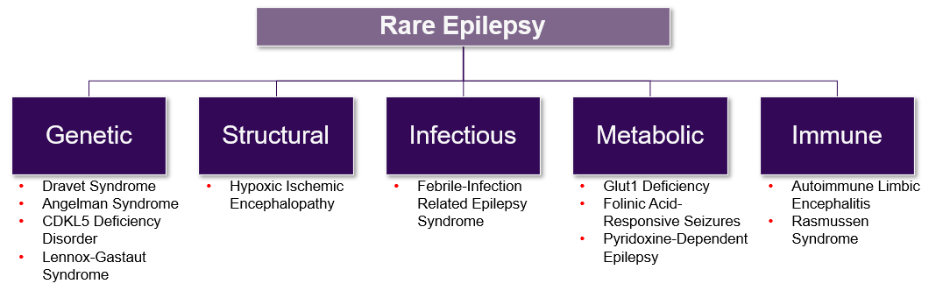
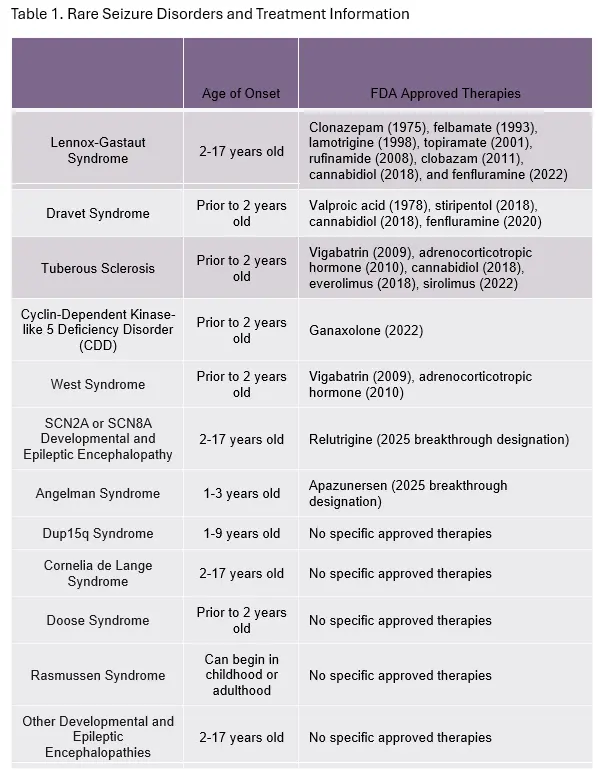
Rare seizure disorders have a complex etiology which may be impacted by factors such as genetic background, anatomical and physiological state, and infectious disease status (Figure 1). This leads to challenges in diagnosing these disorders and developing safe and effective therapeutic agents. Table 1 provides examples of rare seizure disorders, the age of their onset, and the current FDA approved therapies for each.
Diagnosis of rare epilepsy may occur after multiple years of uncontrolled seizures, and symptomology may vary across individuals with a single condition and overlap across disorders. Despite these obstacles, companies are investing in clinical studies to evaluate novel therapeutic agents and gaining marketing approval for products to treat rare epilepsies.
Developing Therapeutic Agents for Rare Seizure Disorders
Given the complexity of etiology and diagnosis for rare seizure disorders, developing safe and effective therapeutics is not always straightforward. These conditions can evolve throughout the life of patients, may express multiple phenotypes/genotypes, can involve severe symptoms, and may progress from one disorder to another. For example, the therapeutic and safety profile of potential drugs change as the brain develops from birth to adulthood, which is particularly relevant as many seizure disorders affect young children. While the physical blood brain barrier is considered mature at birth, the activity of various transporters is modulated according to the needs of the developing brain, with many key drug transporters upregulated to provide nutrients. This may lead to greater neurotoxicity risk (Schmitt 2017). The therapeutic potential of products must be carefully evaluated given that multiple phenotypes and genotypes may be expressed by patients, particularly in individuals diagnosed with disorders such as Dravet’s Syndrome (DS) and Cornelia de Lange Syndrome. Patient treatment may also be complicated by severe symptoms such as sudden unexpected death in epilepsy and developmental delays; as well as the progression of one disorder to another (e.g., initially diagnosed with Dup15q Syndrome followed by Lennox-Gastaut Syndrome [LGS]; or initial diagnoses with West syndrome followed by LGS). Despite these complications, the future for developing therapeutic agents for treating rare seizures is promising given the evolution of the current regulatory and clinical landscape and the potential future directions for this area.
Landscape Analysis: FDA-Approved Therapies for Rare Seizure Disorders
The regulatory and clinical landscape for rare seizure disorders has advanced in recent years, as demonstrated by several FDA approvals for monotherapies, ongoing clinical studies evaluating a range of products in patient populations, and refinement of clinical approaches.
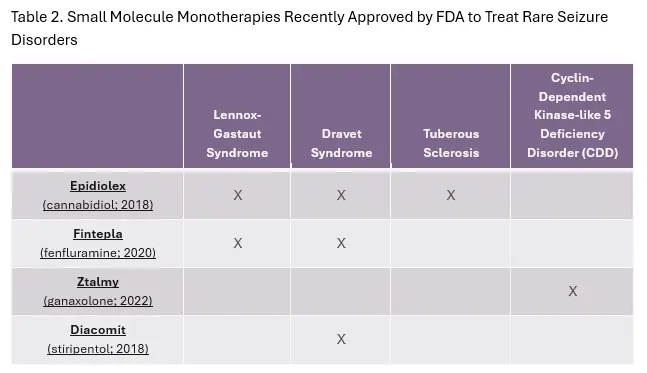
Between 2018 and 2022, the FDA has approved four small molecule monotherapies to treat rare seizure conditions including Lennox-Gastaut Syndrome, DS, Tuberous sclerosis, and Cyclin-dependent kinase-like deficiency disorder (Table 2). Although these small molecules are intended to treat disorders with a genetic etiology, the mechanism of action for these products is not fully understood.
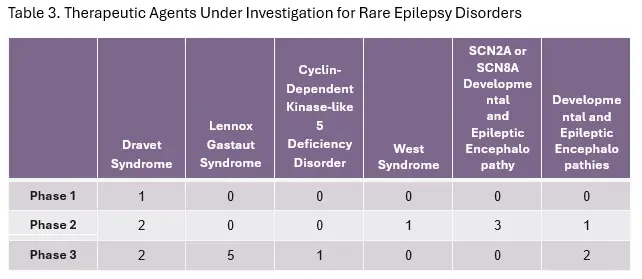
As of the publication of this blog, there are 18 ongoing US clinical trials being conducted to evaluate products (including gene therapies, oligonucleotides, and traditional small molecules) as Phase 1, 2, or 3 studies for rare seizure indications based on data from www.clinicaltrials.gov (Table 3). These and other studies are being refined to better understand rare seizure disorders by defining the full disorder spectrum, addressing comorbidities, characterizing disease progression, and generating real world data. More resources are also being invested in precision medicines.
Together, these efforts have led to increased treatment options for patients and improved clinical programs to develop therapies for rare seizure disorders.
Future Development Directions: Emerging Treatments for Rare Seizure Disorders
Given limited resources and the related costs to develop these products, non-animal models have been created to evaluate potential therapies for rare seizure disorders. Successful non-animal models include patient-derived iPSC assays (Sabitha 2021) and human neuroblastoma SH-SY5Y cells with CDKL5 deletion (Loi 2020). Additionally, pharmacokinetic/pharmacodynamic (PK/PD) modeling can help reduce or eliminate the need for animal models, reducing cost and streamlining the development of drugs for rare seizure disorders. Mechanistic models (i.e., physiologically based pharmacokinetic modeling, quantitative systems pharmacology) can predict PK exposures in the brain (determining the ability and extent of drug penetration of the blood brain barrier). Modeling techniques can also be used to optimize study designs, select dose, and predict dose adjustments needed for specific populations (e.g., pregnant women, patients at the extremes of age, and patients with organ impairment).
Additionally, there is growing interest in plant-derived agents such as cannabinoids as new treatments. Epidiolox (approved in 2018) is the first FDA approved cannabidiol product and serves as treatment for Lennox-Gastaut syndrome, DS, or tuberous sclerosis complex. Cannabinoids may be obtained from cannabis plants, which may lower production costs compared to synthetic products. There are multiple non-clinical studies that demonstrate the potential efficacy of CBD in models for many rare seizure disorders. CBD was evaluated with animal models of CDD and Angelman Syndrome and both disorders showed signs of improvement following treatment (Gu 2019, Li 2024). In zebrafish models of DS, additional cannabinoids (Delta9-THC, cannabinol, and linalool) demonstrated reduction in seizure-like activity (Thornton 2020), although mouse models of DS yielded variable outcomes based on mode of seizure induction and mixture of cannabinoids (Benson 2020).
Although cannabinoids have demonstrated efficacy for various indications, these drugs have notable (although manageable) toxicities. In particular, there is evidence that Delta9-THC produces biphasic effects in seizure frequency. At low doses, seizures are prevented or reduced, but at doses above the regulatory approved threshold, seizures increase. Extensive clinical use history may be used to determine dosing and ensure safety (Murray 2024). Examples of biphasic effects shed light on the complex mechanism of action of cannabinoid products in seizure management and highlight the need for continuing research.
Conclusions
The use of non-animal models to evaluate therapeutics and the development of plant-derived therapies to treat rare seizure disorders both offer unique cost-reduction potential for researching and developing future therapeutics. Harnessing both non-animal models and plant-derived products in the same development program provides the opportunity for synergy. Non-animal models may decrease in vivo evaluation time of candidate drugs such as cannabinoids and PK/PD models may clarify exposure profiles and the mechanisms of action, especially in situations in which limited data are available to make safety decisions (e.g., cannabinoids in pediatric patients). Innovations related to these models and reductions in costs and time are critical to address unmet medical need in patients with rare seizure disorders.
At Allucent, our experts integrate these approaches into practical solutions for rare disease development programs. To learn more about our expertise in helping small to mid-size biopharma bring rare disease therapies to light, visit our website: Orphan and Rare Diseases CRO Services | Allucent
References
Benson MJ, Anderson LL, Low IK, et al. Evaluation of the Possible Anticonvulsant Effect of Δ9-Tetrahydrocannabinolic Acid in Murine Seizure Models. Cannabis and Cannabinoid Research. 2020;7(1):46-57.
Gu B, Zhu M, Glass MR, et al. Cannabidiol attenuates seizures and EEG abnormalities in Angelman syndrome model mice. J Clin Invest. 2019;129(12):5462-5467.
Li X, Yennawar M, Wiest A, et al. Cannabidiol attenuates seizure susceptibility and behavioural deficits in adult CDKL5(R59X) knock-in mice. Eur J Neurosci. 2024;59(12):3337-3352.
Loi M, Trazzi S, Fuchs C, et al. Increased DNA Damage and Apoptosis in CDKL5-Deficient Neurons. Mol Neurobiol. 2020;57(5):2244-2262.
Murray CH, Gannon BM, Winsauer PJ, Cooper ZD, Delatte MS. The Development of Cannabinoids as Therapeutic Agents in the United States. Pharmacol Rev. 2024;76(5):915-955.
Sabitha KR, Shetty AK, Upadhya D. Patient-derived iPSC modeling of rare neurodevelopmental disorders: Molecular pathophysiology and prospective therapies. Neurosci Biobehav Rev. 2021;121:201-219.
Schmitt G, Parrott N, Prinssen E, Barrow P. The great barrier belief: The blood-brain barrier and considerations for juvenile toxicity studies. Reprod Toxicol. 2017;72:129-135.
Thornton C, Dickson KE, Carty DR, Ashpole NM, Willett KL. Cannabis constituents reduce seizure behavior in chemically-induced and scn1a-mutant zebrafish. Epilepsy Behav. 2020;110:107152.